Viral nutrition: new study reveals microbes nourished by consuming viruses

John P. DeLong, James L. Van, Zeina Al-Ameeli, Irina V. Agarkova and David D. Dunigan, ‘The consumption of viruses returns energy to food chains’, Proceedings of the National Academy of Sciences, December 27, 2022. https://doi.org/10.1073/pnas.221500012
At various peaks of the COVID-19 pandemic, viruses have had a reputation as destroyers of public health systems and human lives. They have a peculiar biology — while inert outside a living body , but inside, they hijack the cellular machinery to feed, replicate and spread. This association with disease and death has come to define their form in the public imagination, redeemed not even by the fact that there are other microbes that destroy viruses. But a new study, published on December 27 by researchers at the University of Nebraska-Lincoln, offers to upend this.
The authors of the study have reported that a particular genus of plankton can consume viruses as well as “grow and divide given only viruses to eat”. We already know of other cells that can ‘consume’ viruses in an effort to destroy them — such as the macrophage cells of the human immune system.
The difference lies in being able to ‘eat’ viruses to fulfil one’s biological imperatives.
Plankton of the genus Halteria, they claim to have found, can each consume 10,000 to a million virus particles a day, increase their population using the metabolised energy, and provide more food for the zooplanktons that consume the Halteria. This could be significant for the marine food chain.

Halteria grandinella.
| Photo Credit:
Picturepest/Flickr
What are plankton?
Plankton are microscopic organisms that can only move with a current. They don’t have any facilities to actively propel themselves. Halteria plankton are ciliates, meaning they have hair-like structures called cilia on their surface. Sometimes they can beat some of these cilia to jump short distances, but not often as it they can’t do this often because it requires too much energy.
What do plankton do in the food chain?
A type of plankton — the phytoplankton — is found nearer the surface of many water bodies. It is an autotroph, which means it can make its own food which it does by consuming carbon dioxide, among other compounds, through photosynthesis. Small fish and larger plankton called zooplankton eat phytoplankton for their nutrition; they are in turn eaten by larger fish, and so forth.
When phytoplankton die, they drift around where they are, becoming part of a coastal nutrient cycle, or they drift down towards the seafloor, where they decompose. Their constituents then become available for microbes or are sequestered into the seafloor.
So, phytoplankton bring carbon and other nutrients from the atmosphere and sea surface down to the seafloor and help replenish the food chain (and also ‘trap’ carbon into their own bodies and as sediments). They are joined by bacteria that make their own food by oxidising sulphur, iron or hydrogen, in a process called chemosynthesis.
The role of Halteria
Halteria plankton are found in large numbers in freshwater bodies. They are heterotrophs meaning they can’t produce their own food. Instead, they are well-known bacterivores — they consume bacteria to power themselves.
In the words of a noted 1999 paper, viruses “short-circuit” the process of nutrients moving up the food chain. They infect and kill both bacteria and plankton, releasing organic matter that dissolves in the water. Note that scientists have modelled this ‘viral shunt’ and studied it in the lab; investigations of its real-world impact are ongoing.
In the new study’s paper, the authors wrote that by also consuming viruses for nutrition, Halteria plankton can recover the nutrients lost in the viral shunt and bring them back into the food chain. “This flow would depend on virion size and nutritional content, which varies among strains,” they added, “but it is already clear that viruses of a wide range of sizes can be taken up”.
Redemption for viruses?
Viruses are the single-most populous life-form on Earth; their cumulative biomass outweighs that of humans by a factor of 10^10. In 2020, Curtis Suttle, a co-author of the 1999 paper, called them “fantastic nutritional sources”.
The finding wasn’t unexpected. In 2020, a group from the U.S. and Spain published a paper after 10 years of data-taking and study, positing that some protists (eukaryotes that aren’t plants, animals or fungi; eukaryotes are organisms whose cells have nuclei) might be munching on viruses. They wrote that their findings “indicate that the viral shunt is complemented by a viral link in the marine microbial food web”.
There have been papers discussing this link since at least 1992.
In the new study, the researchers found that Halteria plankton reduced the population of chloroviruses ‘fed’ to them while growing their own numbers — whereas Paramecium ciliates consumed the chloroviruses but didn’t proliferate. That is, to quote from their paper, “not all ciliates can grow on chloroviruses in these conditions, even when they consume them”.
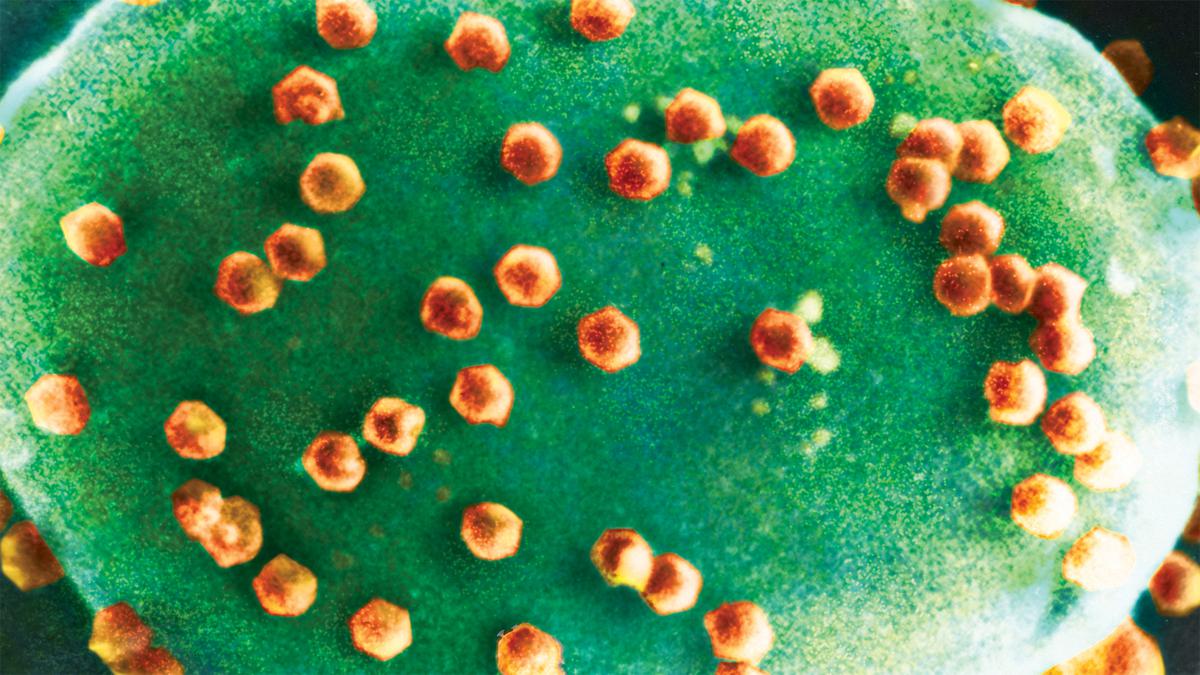
Chlorovirus particles on algae.
| Photo Credit:
Kit Lee and Angie Fox/University of Nebraska-Lincoln
Second, as the population of viruses and Halteria changed over the course of their interaction (in the lab), the researchers used a modelling tool to understand if they fit what we already know about predator-prey interactions in the wild. The model indicated that they did, but John DeLong, a biologist and the lead researcher, said in a press release: “Now, we have to go find out if this is true in nature.”
DeLong et al. also wrote that they don’t yet know how Halteria feeding on viruses could have affected viral evolution.

